Batteries
Maintenance
by John Joyce
Health Warning
Battery electrolyte (sulphuric acid) is extremely corrosive. It will eat its way through most things (including metal, wood, clothes, people) given time. Avoid getting it on your clothes on pain of finding holes in them next time you wash them! On skin it starts to sting after a minute or so, but can be washed off with little ill effect, although experience shows that it is rather more unpleasant if it finds a cut. Don't try making up your own electrolyte from neat acid unless you know what you're doing; neat acid is very dangerous to the uninitiated. Note also that the charging process generates quantities of hydrogen and oxygen; theoretically these can explode if a spark or naked flame is nearby but I've not managed it (yet).
Tools Required
Digital multimeter - these are much more robust and accurate than analogue ones. Hydrometer - for checking electrolyte strength. I bought a cheap one from Halfords (they should be next to the batteries!), and it has proved a very useful acquisition. Demineralised /deionised / distilled water - for topping up. Spanners (preferably insulated!).
Electrolyte Level
Electrolyte should only be topped up with demineralised / deionised or distilled water; tap water (soft or hard) will shorten battery life. The damage isn't obvious but the cells will fail prematurely. Use dedicated plastic storage and topping-up containers to avoid contamination; a plastic milk bottle or similar is handy for the latter.
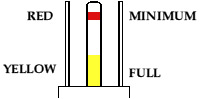
Typical electrolyte level float. The red band indicates 'minimum' when it is about to disappear; the yellow area indicates 'maximum when it appears. This leaves enough space in the cell for it to gas during use without splashing electrolyte everywhere.
The electrolyte level needs to be above the top of the plates and some way below the top of the case - too high and it will splash out during charging and when the vehicle is moving; too low and the battery will lose capacity as the plates become exposed. Most types have some form of indication of maximum / minimum indication, earlier types having a float and later types having markings under the filler cap.
Black rubber cells (BRA2 type) and the early plastic cells (the ones in wooden crates) have floats which have markings for maximum and minimum. Do beware, as occasionally the float will stick or even break; this tends to become obvious when the battery is full and the float still hasn't moved! Later plastic cells (with the flip-cap filler) have a plastic moulding inside the filler neck, with the bottom of the moulding being 'minimum' and the ridge part way up being 'maximum'. These can be filled without removal from the battery box with a bit of ingenuity - use something clean and plastic (eg a drinking straw) as a 'dipstick'.
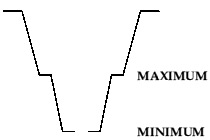
My interpretation of the markings on the flip-cap type filler; the bottom of the plastic moulding is 'minimum' and the ridge part way up is 'maximum'. If anybody knows different then do let me know!
A final word - top up batteries before charging them, not afterwards. The water will tend to sit on top of the existing electrolyte, and in frosty weather it can then freeze and split the case. Charging for a few hours will cause the batteries to gas a little and mix it in.
Electrolyte Strength
This should be checked from time to time, depending on the usage cycle of the batteries. It can also be checked more often to give some indication of whether a particular usage cycle is keeping them charged sufficiently. A further use is for identifying failing cells. (see 'fault finding', below) N.B. Don't try to use a hydrometer straight after topping up; as noted above, the water tends to sit on top for a while. Charge until the cells have been bubbling for an hour or two.
Take a sample of electrolyte from a cell; sufficient so that the float actually moves, but not so much that it hits the top of the hydrometer. Hold the hydrometer vertically and tap it gently to settle the float, but beware of acid dripping from the nozzle in the process. A reading of 1.250-1.275 seems to be a typical result for a charged cell, at least with the hydrometer I have. Below 1.225 and charging is required or the electrolyte is weak; below 1.150 and the cell is flat or failing. Of particular interest is differences between cells or over time.
Charging
The basic instruction is a day's charging every month or two should keep a healthy set of batteries in good shape. If they are in regular use on a trailer or generator-fitted power car then more regular recharging will be needed, perhaps even daily if heaters are in use. Alternator-fitted power cars should need no charging provided that they are used reasonably often. The worst thing to do is to undercharge.
Chargers need careful selection - ignore the 'boost' rating as this is largely irrelevant. A continuous output current capability of 30amps is satisfactory. Machine Mart offer (or at least, used to offer) a reasonably pricesd suitable one (Rally 250 or something like that).
All kinds of arrangements are made for charging, with leads of one kind or another, often made with inadequately sized cable. The rule here is that the longer the charging cable, and the smaller the cable diameter, the longer it will take to charge properly. It is much better to take the charger to the vehicle with mains extension leads as problems with voltage drops are much less severe. If running long leads, aim for 25mm^2 (ie welding cable size) for at least the majority of the cable, perhaps with a short length of thinner cable to actually connect to the vehicle. Although the current is easily handled by smaller cable, it will have a substantial voltage drop which will slow charging down.
Connections
Check all connections in both battery boxes, the battery switch / fuse box ("BIS box" or "Electrical Control Box") are tight. If in doubt, remove, clean, and reassemble tightly with a thin smear of grease on everything. It is wise during overhaul to spend a couple of hours doing this to all the connections. Look out for corrosion on the cell terminals inside the battery boxes, evident as a white or blue powder. This is caused by a little of the acid in the cells escaping during charging, corroding anything nearby. Clean the affected parts thoroughly and smear lightly with grease to prevent a re-occurrence.
Renovating Batteries
A problem which faces many owners is that a newly-acquired vehicle has been left to stand for months or even years, and the batteries are both well and truly flat, and reluctant to recharge. The advice in this case is simple: top up the electrolyte, attach a battery charger as soon as possible, and leave for a week or two. DMU batteries are remarkably resilient to being left for a long time, and will usually recover reasonably well. However, charging for odd days perhaps once a week when the owner is around is not a satisfactory substitute - if a continuous mains supply cannot be arranged for a week or two, take them off and recharge elsewhere.
Storage
New cells are fully charged, then emptied and sealed under a vacuum; they can be stored for years. Adding electrolyte produces a fully charged cell. This is difficult to do for most people. Emptying the electrolyte out from a fully-charged cell is unwise, as the plates will react with air which will destroy the active material. It has been suggested that discharged cells can be emptied and sealed, but I have not tried this - at a guess the sulphate coating on the plates will harden and recharging will become difficult.
Consequently I would recommend storage somewhere that recharging is easy, with a charger connected perhaps one day every month or two. We have stored some for several years and they appear not to be deteriorating badly.


